Last year I wrote about a new service that aims to apply crowdsourcing to improve medical trials. Snazzily titled, the Platform for Engaging Everyone Responsibly (PEER) will allow individuals to share their health information using a comprehensive dashboard. PEER engages with participants by asking them to complete a gamified survey that gives them real time information against other users on the site.
This information will then be used to improve the clinical trials process, both in terms of cost and efficiency. While the rest of the developed world recruits individuals for various endeavours through customized and engaging online tools, biomedical research still tries to enroll individuals as it has for decades: leaning on clinicians who have limited time, posting ads on buses and in subways, even newspapers.
The aim is to ensure that the right clinical trial is directed at the specific person that requires it, rather than relying on chance to match up the respective parties. What’s more, it also ensures that any data generated from the trials is shared with other researchers, thus removing the proprietary silos that often form around research efforts.
New findings by Lilly COI underlined the importance of such an improvement. They have explored how aware cancer patients are of any clinical trials that they could be participating in, and found that just 16% of cancer patients were aware of any relevant trials operating at the time they were considering treatment. This was despite a whopping 95% of patients expressing a desire to consider such an option should they be made aware of it.
Hopefully projects such as PEER will help to raise awareness of the trials that are in operation, whilst also greatly improving the drug development process. You can see the results of the Lilly COI study in the infographic below.
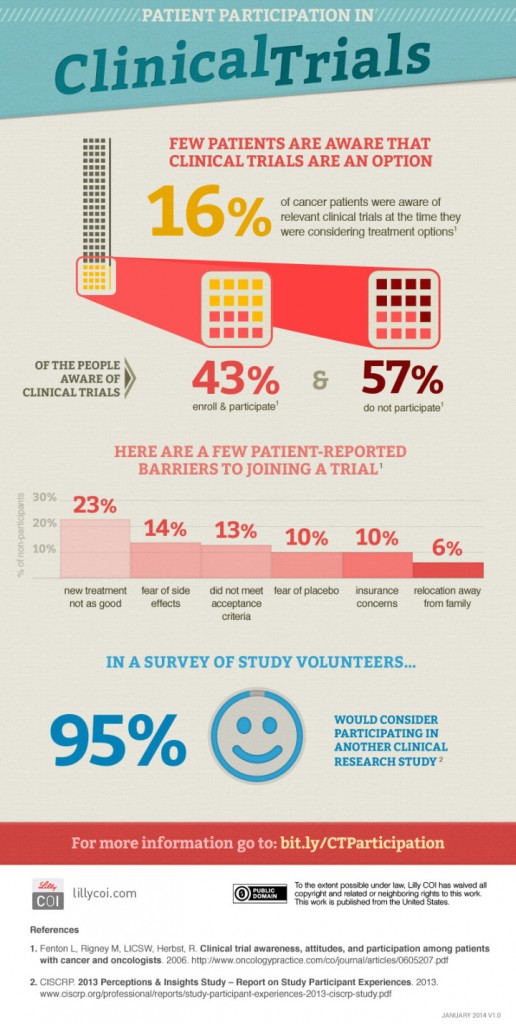
That 16% figure is awful. What a waste.
Interesting piece here on this topic
http://www.nejm.org/doi/full/10.1056/NEJMe1400850
In the fall of 2013, the Institute of Medicine (IOM) convened a committee, on which I serve, to examine the sharing of data in the setting of clinical trials. The committee is charged with reviewing current practices on data sharing in the context of randomized, controlled trials and with making recommendations for future data-sharing standards. Over the past few months, the committee has prepared a draft report that reviews current practices on data sharing and lays out a number of potential data-sharing models. Full details regarding the committee’s charge and the interim report are available at http://www.iom.edu/activities/research/sharingclinicaltr....
Open-data advocates argue that all the study data should be available to anyone at the time the first report is published or even earlier. Others argue that to maintain an incentive for researchers to pursue clinical investigations and to give those who gathered the data a chance to prepare and publish further reports, there should be a period of some specified length during which the data gatherers would have exclusive access to the information. Since these researchers could always agree to collaborate with others who were not involved in the study in order to use the data to help answer a scientific question, the period of exclusivity would really apply only to noncollaborative use of the data. That is, there would be a defined period during which the data would not be available to those who wanted to perform their own analyses and draw conclusions that could, for example, provide them with a scientific or commercial competitive advantage over the researchers who had originally gathered the data or allow them to derive conclusions that are potentially at odds with those drawn in the original publication.
As members of a community that either produces or uses data, what approach do you think serves our community best? There is no need to reply to the Journal, but please read the interim report and let the IOM know how you feel about this and the many other critical issues related to data sharing that are reviewed in the document. The IOM is collecting comments until March 24, 2014, at www8.nationalacademies.org/cp/projectview.aspx?key=49578.”
Hospital or other health care facilities should introduce clinical trials to patients, for most cancer patients are not aware of what a clinical trial is and how it can help them with their recovery.